usp class vi vs iso 10993
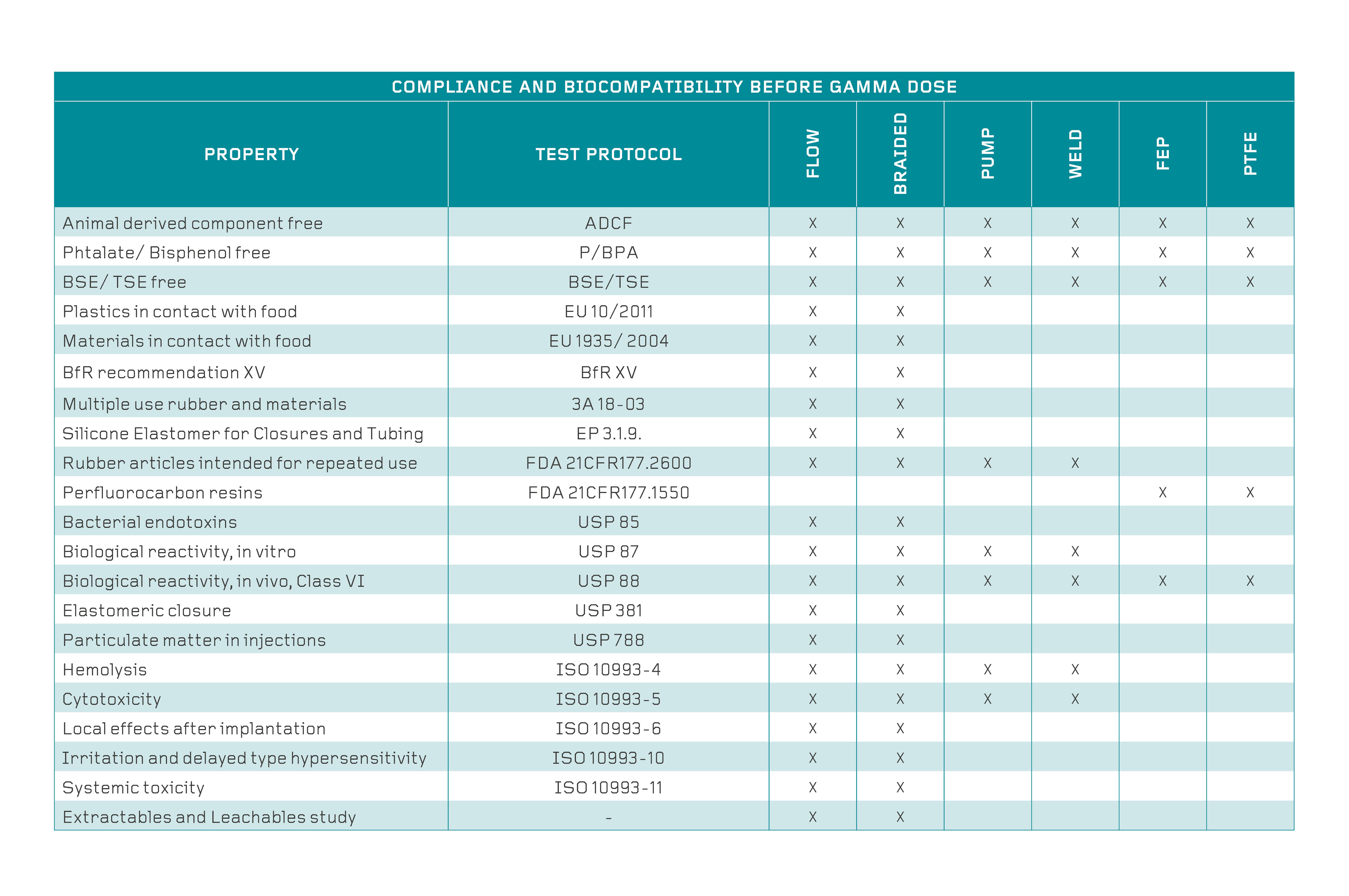
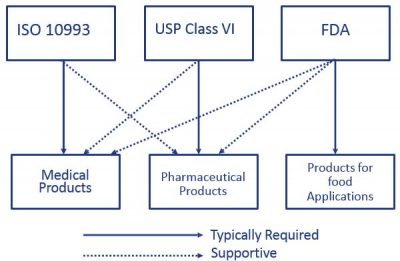
For most patient-contact applications a material that meets US Pharmacopeia USP Class VI andor ISO 109933 will be required. Biocompatibility testing biocompatible materials biocompatible rubber ISO 10993 medical molder medical molding medical silicones USP Class VI.

What Is Iso 10993 How Is It Different From Usp Class Vi Ppt Download
The guidance memo wasis G95-1.
. A more rigorous standard for the biological evaluation of medical devices is ISO-10993. Class VI and ISO 10993 are recommendations for testing based on the use of the final device. ISO-10993 is a standard that utilizes systemic toxicity and intracutaneous reactivity testing.
A selection of Figure 4 VisiJet Accura and DuraForm plastic materials have met the requirements of ISO 10993-5 -10 or USP Class VI testing. If yes to the first question then USP Class VI is not a relevant qualification for it. Take an ASTM D2000 call out.
So does ISO 10993. A number of our plastic materials are ISO-10993 or USP Class VI capable. The most stringent Class VI requires three types of tests.
This post will take a deeper look at what biocompatibility is and how it is defined by the International Standards Organization. Depending on the devices use the sterilization process might obligate you to do ISO 10993. A rubber compound has set physical parameters it needs to meet.
You might establish biocompatibility via making the device of a Recognized Consensus Standard material using a validated process that does not degrade that material or by ISO 10993 testing. While it is possible a USP Class VI material could also be ISO 10993 compliant its not a given and USP Class VI alone is not sufficient for adherence to ISO 10993. Unlike other rubber standards theres no one standard that engineers use for an approval.
USP Class VI vs. The materials listed below are ideal for. That said the lack of risk assessment in USP Class VI can be a problem.
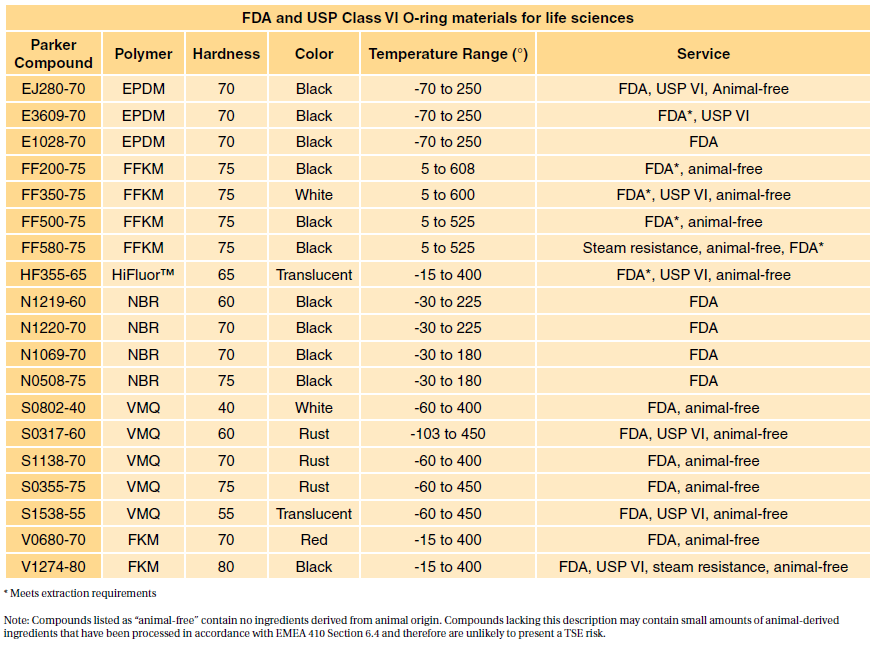
USP class qualification no longer plays any role in medical device materials evaluation. In fact USP Class VI is sometimes seen as a minimum requirement for biocompatibility. Medical Silicone Rubber Molding and Silicone Rubber Mold Materials.
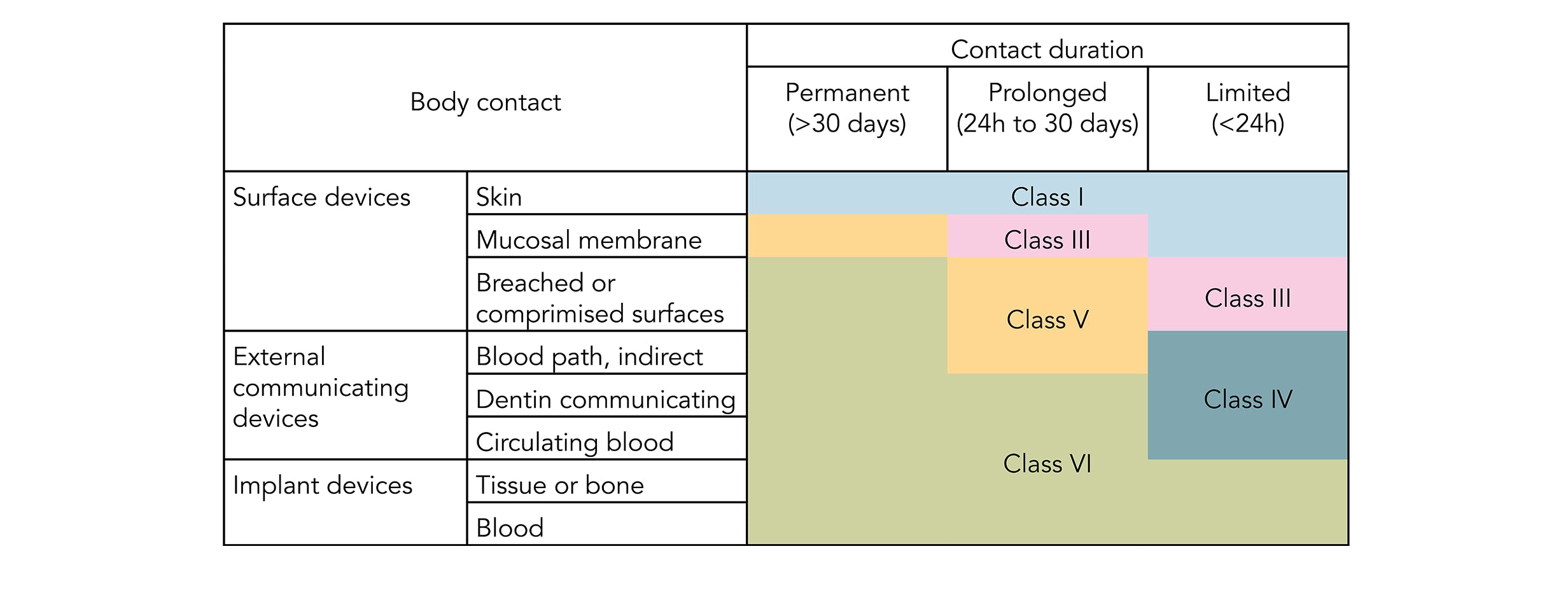
Parylene has a long history of use as a protective coating for medical device biocompatibility and conforms to the USP Class VI and ISO 10993 standards. Testing Testing to the highest ISO-10993 standards can add months of time and be very costly according to the Medical Device Testing Guide by Toxikon Inc. ISO 10993 is designed for medical products that remain permanently or for a very long time in the human body so for shorter applications a USP Class VI or even a lower USP Class certification is often sufficient.
That being said if you cant get an ISO 10993 compliant material often because the material simply hasnt been tested using a USP Class VI material is a less risky option. USP Class VI Testing is only one standard of biocompatibility however. USP Class VI Regiment Irritation Systemic Injection Implantation 1 week Biological Evaluation Plan BEP.
Biological evaluation of medical devices Part 1. In fact USP Class VI has been largely superseded since the release of ISO 10993 in 1995. In 1995 the FDA adopted ISO 10993 as its biocompatibility approach.
USP Class VI demands an intracutaneous irritation test. Predictable Safe and Stable. 3D printing of dental and orthopedic surgical guides.
USP Class VI. Typically the terms USP Class VI or ISO 10993 materials are used. Though not a limited series of tests some biocompatibility requirements for medical devices may exceed the testing performed in USP Class VI.
For this reason the FDA provides a standard 21 CFR1772600 defining allowable rubber compound ingredients and extractibles based on toxicity and carcinogenicity. Many medical device companies are familiar with USP Class VI but that standard isnt as strict as ISO 10993. Depending on the devices application and how the cable components will interact with the patient this higher level testing may not be needed.
Duration similar to ISO-10993 and each class has a different set of testing requirements. To begin let us address just what biocompatibility is. This is their current stance today.
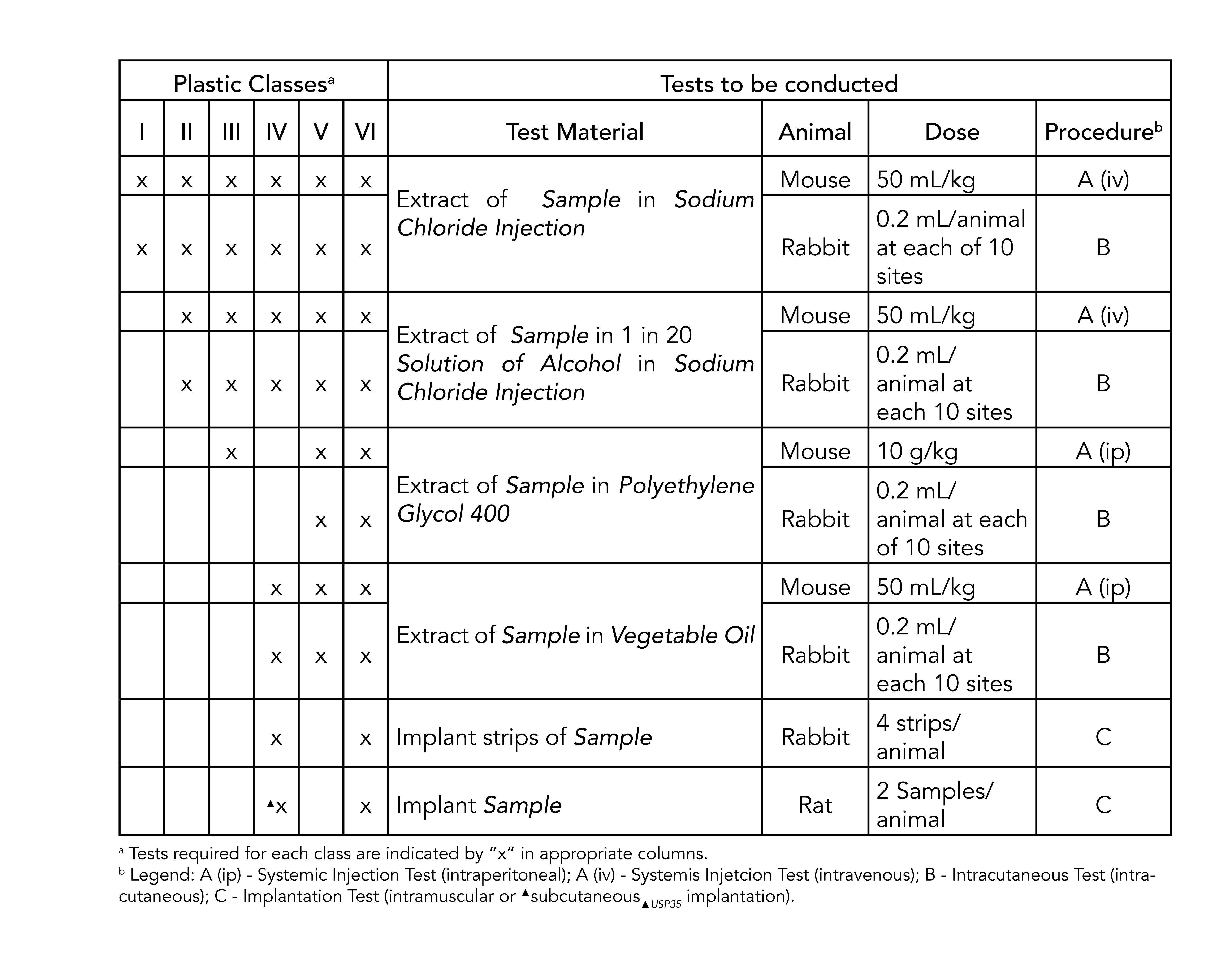
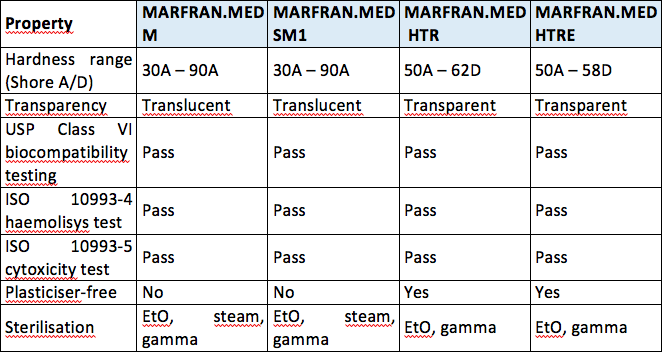
USP Class VI testing is conducted by producing an extract of the product with different extraction fluids such as polyethylene glycol and vegetable oil and injecting it in specimen rabbits and mice in vivo alive to observe the biological response to the extract. We carry a wide range of materials from the worlds top medical resin suppliers including USP Class VI and ISO 10993 certified biocompatible resins with full FDA Master File support. Sealable and weldable either pre- or post-sterilization C-Flex 072 provides prolonged pump life Sterilizable by gamma irradiation and autoclave Product Validation Test Summaries available upon request Moldable bondable and formable for single-use assemblies and overmolds Temperature.
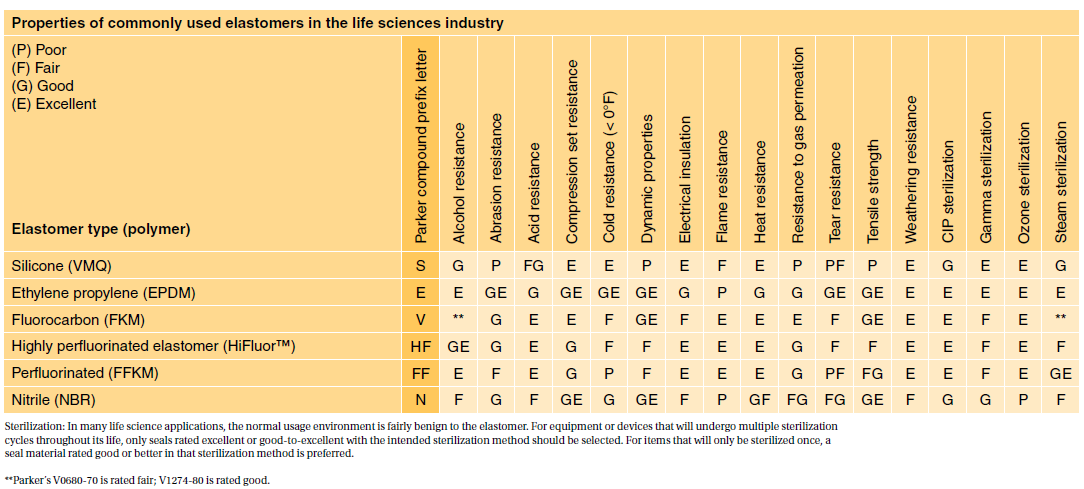
Most applications are fairly benign to elastomers. 3D printing of one day crown prep guides. Then you need to understand the differences between ISO 10993 and USP Class VI and the nature of each standard.
It depends to a large extent on the application and therefore also on the application period of the finished product. USP Class VI ISO 10993-5 Cytotoxicity In-Vitro Features Benefi ts. It is transparent pin-hole free and conforms precisely to any surfaces features.
Evaluation and testing within a risk management process. Testing is commonly done as per USP which requires three types of. Our portfolio approach offers the most expansive selection of medical resin materials in the industry balancing performance cost.
Up-to-date materials manufacturers provide both USP and ISO 10993 test data to support both pharma and device customers. Parylenes thickness is critically controlled and extremely consistent. Systemic injection test Intracutaneous test Implantation test USP standards for the first two tests in the list above are nearly identical to ISO-10993 standards for.
However Class VI also requires subacute toxicity and implantation effects which many ISO 10993 categories do not. The Value Of USP Class VI Testing For Medical Device Cable. Though not a limited series of tests some biocompatibility requirements for medical devices may exceed the testing performed in USP Class VI.
A more rigorous standard for the biological evaluation of medical devices is ISO-10993.

Duraform Pa Certification Usp Class Vi Iso 10993 And Food Contact
Brilliant Mind The World Of Tubing For Medical Use Medical Plastics News

Understanding Food Grade Vs Biocompatibility For Medical Device Materials Medical Product Outsourcing

Usp Class Vi Foster Corporation
Biocompatibility Of Plastics Zeus

Heat Resistant Thermoplastic Ultem 9085 Baltic3d Eu
Usp31nf26s1 C1031 General Chapters 1031 The Biocompatibility Of Materials Used In Drug Containers Medical Devices And Implants
Material Selection Medical Injection Molding Xcentric Mold

Silicones Wacker Silicones For Medical Why Silicones Extremely

What Is Iso 10993 How Is It Different From Usp Class Vi Ppt Download

Iso 10993 Vs Usp Class Vi Medical Molding And Bicompatible Rubber The Rubber Group